 Applications of copper isotopeĬopper provides a relatively large number of radioisotopes that are suitable for use in nuclear medicine. 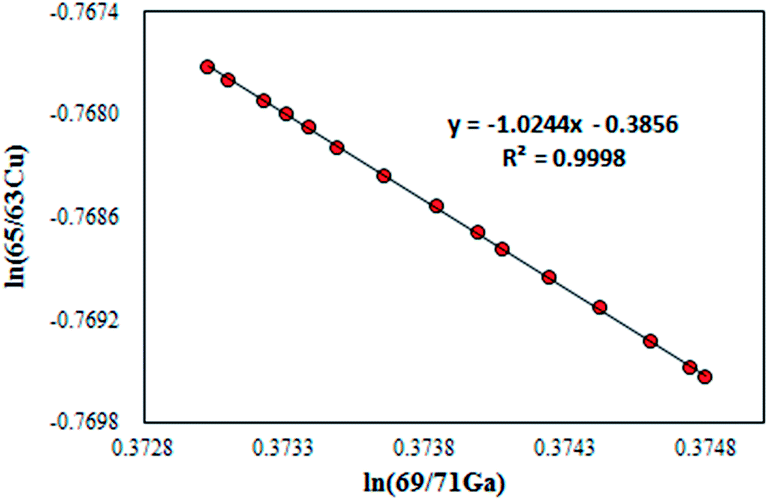
According to the above, copper 65 is less common than copper 63. American Elements is responsible for the production of copper 65 for biological and biomedical labeling, as target materials and other applications and Copper 65 has a density of 7.17. Is copper 63 more common or copper 65? The answer can be said Copper 65 is a non-radioactive and stable isotope which is both produce by fission or naturally occur. We would like to point out that copper 64 radioisotope is one of the most important copper radioisotopes, which emits gamma and beta rays and has a half-life of 12.7 hours (decrease in the activity of this radioisotope) used in studies of tumor metabolism and many types of cancer. Also, copper 63 is useful in producing Cu-64 and is important in cancer diagnosis and treatment. Copper 63 has a density of 7.93 and is used for production of medical radioisotope Zn-62. It’s a non-radioactive and stable isotope which is both produce by fission or naturally occur. You might be wondering is copper 63 radioactive? The answer is no. In the following, we will examine the characteristics of these two isotopes. For example, cu-63 isotope and cu-65 isotope are used to study copper metabolism and gastrointestinal diseases. Copper isotopes 63 and 65 are used in various industries. In the copper element, in addition to a large number of radioisotopes, there are two copper stable isotopes, Cu-63 and Cu-65 and the amount of them in nature are Cu-63 (69.17%) and Cu-65 (30.83%). To answer the question, how many copper isotopes are there? Stay with us. ZTI supplies this product as powder and ingots with 99.999% purity. Both isotopes have the same properties and are both stable. Both Cu-63 and Cu-65 atoms have 29 protons, but Cu-63 has 34 neutrons and Cu-65 has 36 neutrons. How many isotopes does copper have?įor example, copper has two forms: copper 63, which is 70% of all copper in nature, and copper 65, which makes up 30% of the rest. Copper isotopes are one of the most important isotopes. Isotope is a Greek word and refers to forms of atoms that differ in atomic mass. 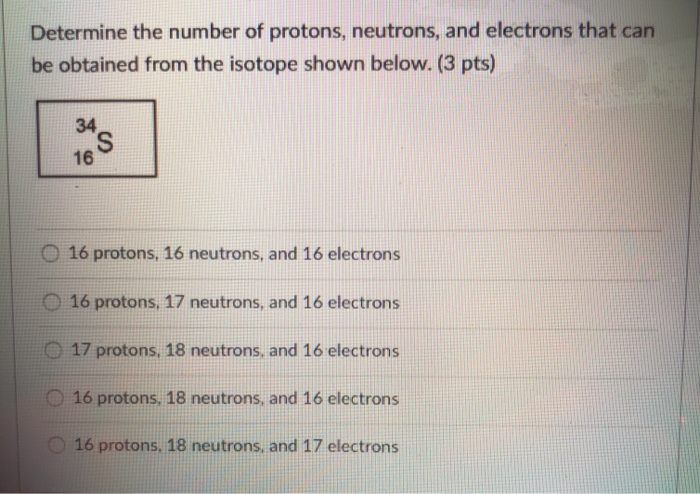
Meanwhile, the term used for atoms with the same atomic number and different atomic mass is called isotope. Atoms and elements are found in nature in different forms, each of these forms has its own characteristics. These things help to recognize the elements and distinction between them. Atomic number and atomic mass of elements are among the most important properties that are studied in different elements and materials. Historically, the use of stable isotopes began in the 1960s for the production of industrial and medical radioisotopes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
